|
The other colors come from different gases such as helium, argon, mercury and combinations with each other and with neon.Trending Questions What is the additive in a black top tube? What do elements in the same group have? If there is 50cc left in glass and the glass holds 150cc you should record the intake as? What happens when a substance sublimes? What is the most common conductor? What is the charge of glucose? Is Magnetic quantum number ever larger than the principle quantum number? What information does the column number provide on the periodic table? What is ionic equation for Na2CO3 plus HCl? What sensitivity sandell? Y shaped regions where the 2 strands of DNA separate? What is a partical in an atom with no charge? What is the maximum number of electrons that can occupy orbital? Acids change blue litmus paper? What stores sell rock salt? What is the ka of the first ionization of h3po4 if you initially had 0. The term "neon sign" is commonly used for all such luminous gas signs, but the neon produces only the bright orange characteristic color. The brilliant emission from low pressure neon gas when it is excited by an electric discharge has made it very popular for illuminated signs. The relative abundance of 20Ne is 90.92 and 21 Ne is. Absolute values for the isotopic abundance ratios of atmospheric neon have been determined by mass spectrometry. The number of neutrons depends on the isotope of the element. Neon-20 has a mass of 19.9924 amu Neon-21 has a mass of 20.9940 amu Neon-22 has a mass of 21.9914 amu. Now, the relative atomic mass of naturally occuring neon ( mass number of isotope. The mass number is a count of the number of particles in an atoms nucleus. The difference between the mass number of the neon atom and the number of protons is ten. The difference between the mass number of the neon atom and the number of protons is ten. Given, Naturally occuring neon is 90.9 neon-20, 0.3 neon-21, 8.8 neon-22. There you can find the metals, semi-conductor (s), non-metal (s), inert noble gas (ses), Halogens, Lanthanoides. Where more than one isotope exists, the value given is the abundance weighted average. This is approximately the sum of the number of protons and neutrons in the nucleus. Relative atomic mass The mass of an atom relative to that of carbon-12. Please note that the elements do not show their natural relation towards each other as in the Periodic system. It provides energy levels and branching ratios for atomic nuclei of the isotope Ne-19 (neon, atomic number Z 10, mass number A 19). Density is the mass of a substance that would fill 1 cm 3 at room temperature. The unity for atomic mass is gram per mol. In our example, kryptons mass number is 84 since its atomic weight, 83.80, rounds up to 84. The lightest chemical element is Hydrogen and the heaviest is Hassium. 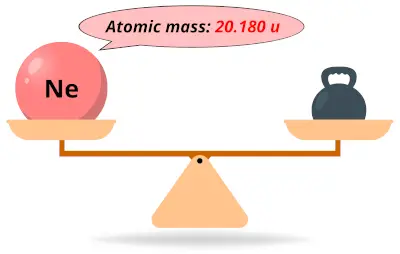
Happily, to find the mass number, all you need to do is round the atomic weight to the nearest whole number. It is isolated mainly by the distillation of liquid air. Unfortunately, the mass number isnt listed on the Table of Elements. It is found in the atmosphere at a concentration of about 0.002%. Get control of 2022 Track your food intake, exercise, sleep and meditation for free. The ground state electronic configuration of neutral neon is He. Molar Mass, Molecular Weight and Elemental Composition Calculator. 
It represents the closing of the n=2 shell of electrons. A container holds 20 g of neon (mass number 20) and also 8 g of helium (mass number 4). Neon atoms have 10 electrons and the shell structure is 2.8.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
